AERATION

ALFAGROG

FISH CARE

HEAT EXCHANGERS

KOI FOOD

PUMPS

TROPICAL FISH

HOSE TAILS

EVOLUTION AQUA

BOWLS
Koi and pond management
We are happy to assist you in all aspects of Koi management. Whether you’re a beginner or an advanced keeper, we are here on hand to help.
We know it can certainly be difficult to maintain Koi ponds, especially if you are just starting out, yet there's nothing better than sitting by your koi pond, admiring your hard work, and relaxing in your very own tranquil sanctuary.
Help & advice
At Birmingham Koi and Aquatics we can offer advice and helpful guidelines about the best equipment for you to use. This can be done either by phone or calling into our shop located in Old Hill, West Midlands - Where we have a comprehensive range of Koi products and aquarium supplies including pond pumps, pond Filter systems, filter media, Koi food, Koi medication and lots more! All at affordable prices... we stock a wide variety of used pond equipment that has been refurbished.

We deliver to your door

Best advice & guidelines

Extensive range of products

All affordable prices
Get in touch or visit us

Birmingham Koi and Aquatics, Unit 1, Elbow Street, Old Hill, West Midlands, B64 6JP

Telephone Number. 07763 226336 POND
Telephone Number. 07762 856229 TROPICAL
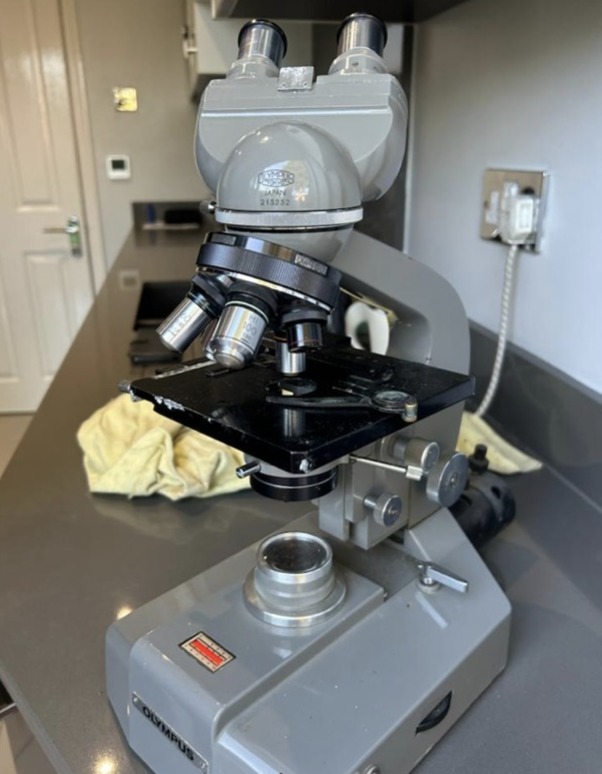
Olympus Microscope
High Quality top of the range.
In very nice condition and works great with a bright light that can be adjusted to your eyes
£350
call 07763 226 336